The Supera™ Peripheral Stent is indicated for the superficial femoral artery (SFA) and the popliteal artery. Engineered by a unique interwoven wire technology, this nitinol stent offers physicians unmatched clinical outcomes across varied lesion complexities and lengths.
Unparalled Flexibility and Zero Fractures
With the Supera™ Stent individual flexible nitinol wires are interwoven for unparalleled flexibility, excellent kink resistance, and the ability to mimic the natural movement of the anatomy. Given the twisting and compression characteristics of the superficial femoral artery and proximal popliteal, this stent is an effective choice. Consequently, data on over 2,000 patients published in 17 studies have shown that at 1 year the Supera™ Stent has zero fractures.

Excellent clinical outcomes
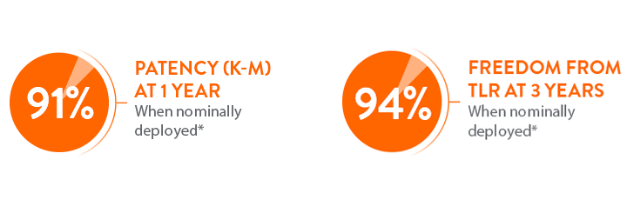
The Supera™ Stent is known for the excellence of its clinical outcomes during percutaneous transluminal angioplasty (PTA) procedures, since this peripheral stent has been studied in more than 2,000 patients and 17 studies worldwide. Supera™ Stent demonstrated excellent 1 yr patency and 3 yr freedom from TLR in the SUPERB trial.
Detailed stent overviews, clinical evidence, cases and more at our partner’s Abbott official website.
Interested? Contact Us:

Gražina




















